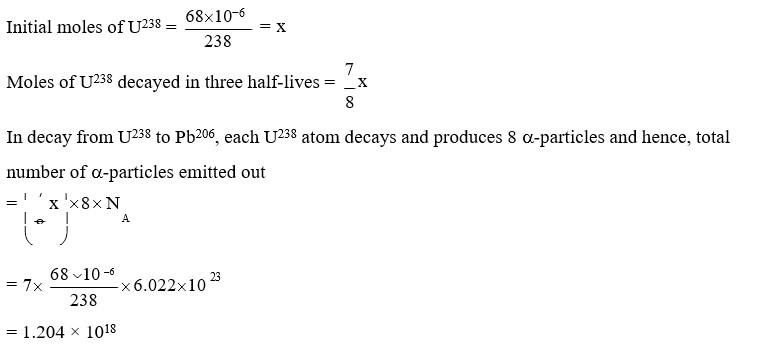${ }^{238}$ Uis known to undergo radioactive decay to form ${ }_{82}^{206} \mathrm{~Pb}$ by emitting alpha and beta particles. A 92 rock initially contained $68 \times 10^{-6} \mathrm{~g}$ of $\stackrel{238}{92} \mathrm{U}$. If the number of alpha particles that it would emit during its radioactive decay of ${ }^{238} \mathrm{Uto}{ }^{206} \mathrm{~Pb}$ in three half-lives is $\mathrm{Z} \times 10^{18}$, then what is the value of $Z$ ?