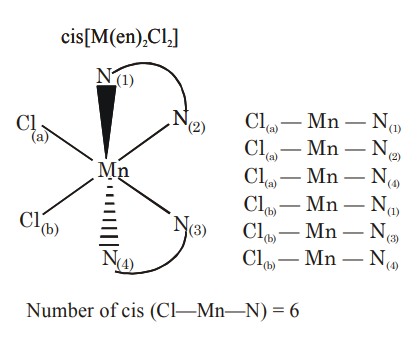
Total number of cis $\mathrm{N}-\mathrm{Mn}-\mathrm{Cl}$ bond angles (that is, $\mathrm{Mn}-\mathrm{N}$ and $\mathrm{Mn}-\mathrm{Cl}$ bonds in cis positions) present in a molecule of cis- $\left[\mathrm{Mn}(\mathrm{en})_2 \mathrm{Cl}_2\right]$ complex is $\_\_\_\_$ (en $=\mathrm{NH}_2 \mathrm{CH}_2 \mathrm{CH}_2 \mathrm{NH}_2$ )