🎉Competishunturns6|Six years of trust. Thousands of JEE dreams guided, together.🎊
🎉Competishunturns6|Six years of trust. Thousands of JEE dreams guided, together.🎊
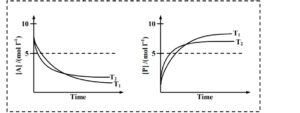 If $\mathrm{T}_2>\mathrm{T}_1$, the correct statement(s) is (are) (Assume $\Delta \mathrm{H}^{\ominus}$ and $\Delta \mathrm{S}^{\ominus}$ are independent of temperature and ratio of $\ln K$ at $\mathrm{T}_1$ to $\ln K$ at $\mathrm{T}_2$ is greater than $\mathrm{T}_2 / \mathrm{T}_1$. Here $H, S, G$ and $K$ are enthalpy, entropy, Gibbs energy and equilibrium constant, respectively.)
If $\mathrm{T}_2>\mathrm{T}_1$, the correct statement(s) is (are) (Assume $\Delta \mathrm{H}^{\ominus}$ and $\Delta \mathrm{S}^{\ominus}$ are independent of temperature and ratio of $\ln K$ at $\mathrm{T}_1$ to $\ln K$ at $\mathrm{T}_2$ is greater than $\mathrm{T}_2 / \mathrm{T}_1$. Here $H, S, G$ and $K$ are enthalpy, entropy, Gibbs energy and equilibrium constant, respectively.)