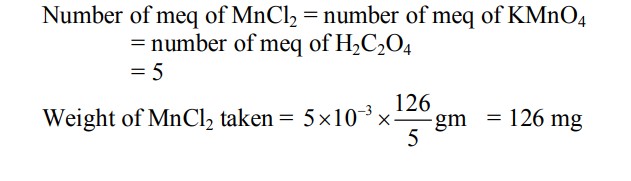To measure the quantity of $\mathrm{MnCl}_2$ dissolved in an aqueous solution, it was completely converted to $\mathrm{KMnO}_4$ using the reaction, $\mathrm{MnCl}_2+\mathrm{K}_2 \mathrm{~S}_2 \mathrm{O}_8+\mathrm{H}_2 \mathrm{O} \rightarrow \mathrm{KMnO}_4+\mathrm{H}_2 \mathrm{SO}_4+\mathrm{HCl}$ (equation not balanced). Few drops of concentrated HCl were added to this solution and gently warmed. Further, oxalic acid ( 225 mg ) was added in portions till the colour of the permanganate ion disappeared. The quantity of $\mathrm{MnCl}_2$ (in mg ) present in the initial solution is $\_\_\_\_$ .
(Atomic weights in $\mathrm{g} \mathrm{mol}^{-1}: \mathrm{Mn}=55, \mathrm{Cl}=35.5$ )