The plot given below shows $\mathrm{P}-\mathrm{T}$ curves (where $P$ is the pressure and $T$ is the temperature) for two solvents $\mathbf{X}$ and $\mathbf{Y}$ and isomolal solu
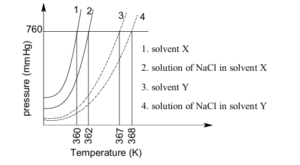
On addition of equal number of moles of a non-volatile solute $\mathbf{S}$ in equal amount (in kg ) of these solvents, the elevation of boiling point of solvent $\mathbf{X}$ is three times that of solvent $\mathbf{Y}$. Solute $\mathbf{S}$ is known to undergo dimerization in these solvents. If the degree of dimerization is 0.7 in solvent $\mathbf{Y}$, the degree of dimerization in solvent $\mathbf{X}$ is $\_\_\_\_$ .tions of NaCl in these solvents. NaCl completely dissociates in both the solvents.
 On addition of equal number of moles of a non-volatile solute $\mathbf{S}$ in equal amount (in kg ) of these solvents, the elevation of boiling point of solvent $\mathbf{X}$ is three times that of solvent $\mathbf{Y}$. Solute $\mathbf{S}$ is known to undergo dimerization in these solvents. If the degree of dimerization is 0.7 in solvent $\mathbf{Y}$, the degree of dimerization in solvent $\mathbf{X}$ is $\_\_\_\_$ .tions of NaCl in these solvents. NaCl completely dissociates in both the solvents.
On addition of equal number of moles of a non-volatile solute $\mathbf{S}$ in equal amount (in kg ) of these solvents, the elevation of boiling point of solvent $\mathbf{X}$ is three times that of solvent $\mathbf{Y}$. Solute $\mathbf{S}$ is known to undergo dimerization in these solvents. If the degree of dimerization is 0.7 in solvent $\mathbf{Y}$, the degree of dimerization in solvent $\mathbf{X}$ is $\_\_\_\_$ .tions of NaCl in these solvents. NaCl completely dissociates in both the solvents.