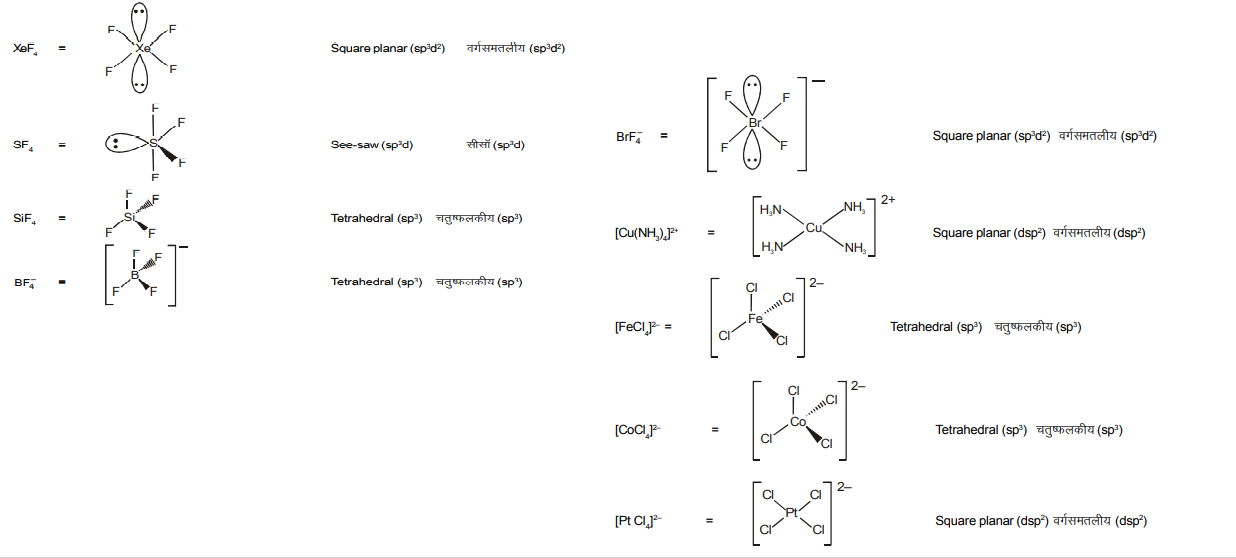
A list of species having the formula $X Z_4$ is given below.
$\mathrm{XeF}_4, \mathrm{SF}_4, \mathrm{SiF}_4 . \mathrm{BF}_4{ }^{-}, \mathrm{BrF}_4{ }^{-},\left[\mathrm{Cu}\left(\mathrm{NH}_3\right)_4\right]^{2+},\left[\mathrm{FeCl}_4\right]^{2-},\left[\mathrm{CoCl}_4\right]^{2-}$ and $\left[\mathrm{PtCl}_4\right]^{2-}$.
Defining shape on the basis of the location of X and Z atoms, the total number of species having a square planar shape is