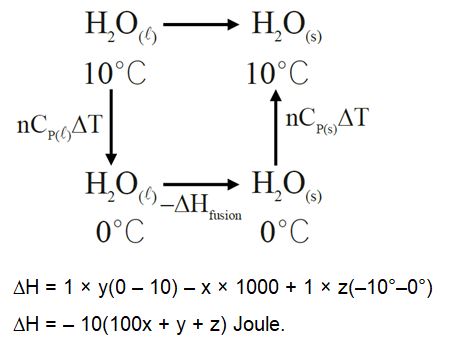
Total enthalpy change for freezing of 1 mol of water at ${10^\circ}{\rm{C}}$ to ice at $ - {10^\circ }{\rm{C}}$ is
(Given: ${\Delta _{{\rm{fus }}}}{\rm{H}} = x\;{\rm{kJ}}/{\rm{mol}}$
${{\rm{C}}_{\rm{p}}}\left[ {{{\rm{H}}_2}{\rm{O}}({\rm{l}})} \right] = y\;{\rm{J}}\;{\rm{mo}}{{\rm{l}}^{ - 1}}\;{{\rm{K}}^{ - 1}}$
${{\rm{C}}_{\rm{p}}}\left[ {{{\rm{H}}_2}{\rm{O}}(\;{\rm{s}})} \right] = z\;{\rm{J}}\;{\rm{mo}}{{\rm{l}}^{ - 1}}\;{{\rm{K}}^{ - 1}}$