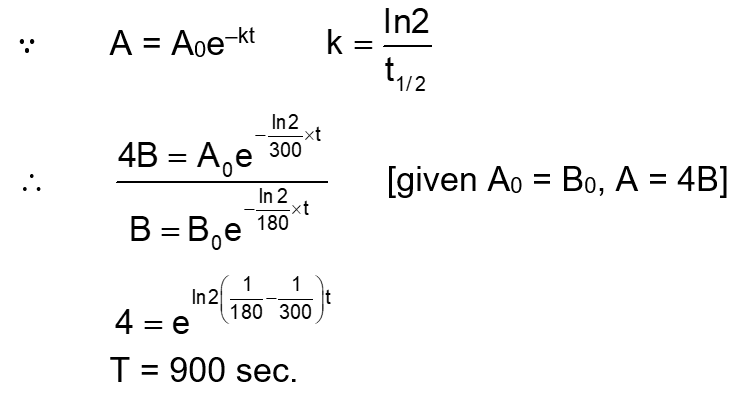
A flask contains a mixture of compounds A and B. Both compounds decompose by first-order kinetics. The half-lives for A and B are 300 s and 180 s, respectively. lf the concentrations of A and B are equal initially, the time required for the concentration of A to be four times that of B (in s) is : (Use In 2 = 0.693)