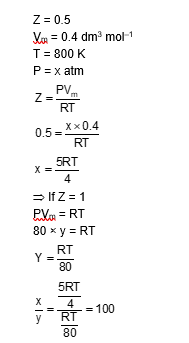A gas has a compressibility factor of 0.5 and a molar volume of $0.4 \mathrm{dm}^3 \mathrm{~mol}^{-1}$ at a temperature of 800 K and pressure $x$ atm. If it shows ideal gas behaviour at the same temperature and pressure, the molar volume will be $y \mathrm{dm}^3 \mathrm{~mol}^{-1}$. The value of $\mathrm{x} / \mathrm{y}$ is $\_\_\_\_$ .
[Use: Gas constant, $\mathrm{R}=8 \times 10-2 \mathrm{~L}$ atm K-1 mol-1]