A graph of vapour pressure and temperature for three different liquids X, Y, and Z is shown below
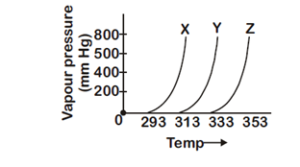
The following inferences are made
(A) X has higher intermolecular interactions compared to Y.
(B) X has lower intermolecular interactions compared to Y
(C) Z has lower intermolecular interactions compared to Y.
The correct inferences is/are
 The following inferences are made
The following inferences are made