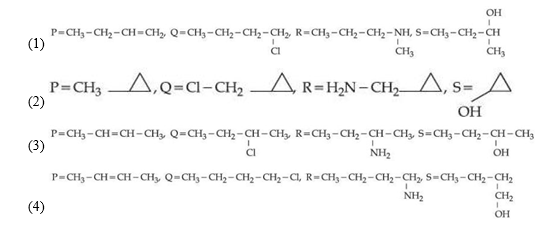
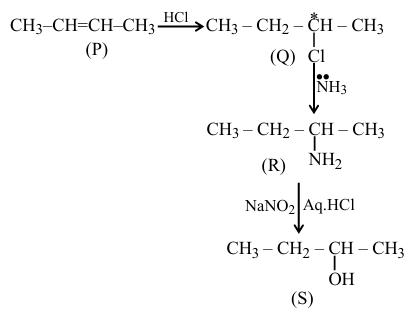
A hydrocarbon ' P ' $\left(\mathrm{C}_4 \mathrm{H}_8\right)$ on reaction with HCl gives an optically active compound ' Q ' $\left(\mathrm{C}_4 \mathrm{H}_9 \mathrm{Cl}\right)$ which on reaction with one mole of ammonia gives compound ' $\mathrm{R}^{\prime}\left(\mathrm{C}_4 \mathrm{H}_{11} \mathrm{~N}\right)$. ' $\mathrm{R}^{\prime}$ on diazotization followed by hydrolysis gives ' $S$ '. Identify $\mathrm{P}, \mathrm{Q}, \mathrm{R}$ and $S$.