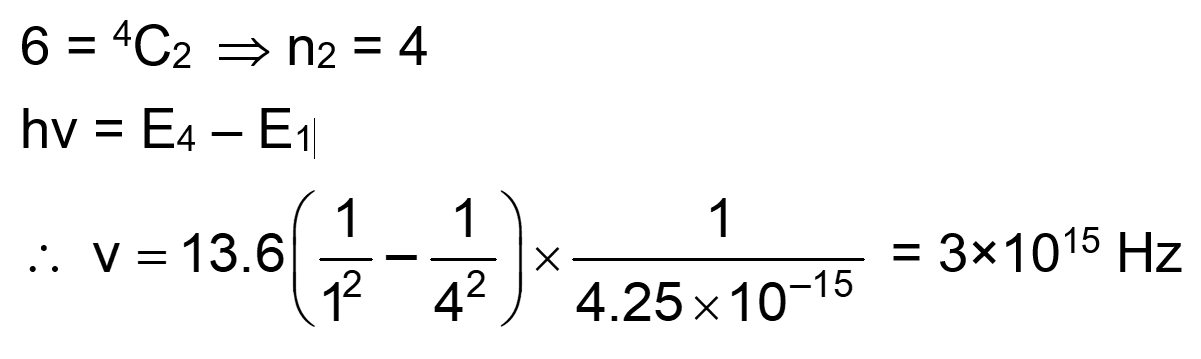A monochromatic light is incident on a hydrogen sample in ground state. Hydrogen atoms absorb a fraction of light and subsequently emit radiation of six different wavelengths. The frequency of incident light is $x \times 10^{15} \mathrm{~Hz}$. The value of $x$ is $\_\_\_\_$ . (Given $\mathrm{h}=4.25 \times 10^{-15} \mathrm{eVs}$ )