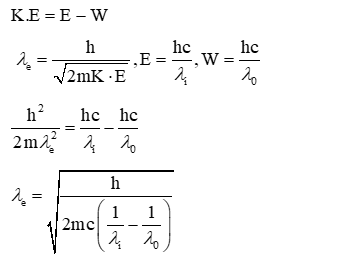A photo-emissive substance is illuminated with a radiation of wavelength $\lambda_i$ so that it releases electrons with de-Broglie wavelength $\lambda_e$. The longest wavelength of radiation that can emit photoelectron is $\lambda_0$. Expression for de-Broglie wavelength is given by : $(\mathrm{m}$ : mass of the electron, h : Planck's constant and c. speed of light)