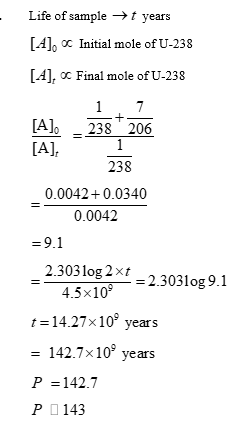A sample initially contains only U-238 isotope of uranium. With time, some of the U-238 radioactively decays into Pb206 while the rest of it remains undisintegrated.
When the age of the sample is $\mathbf{P} \times 108$ years, the ratio of mass of $\mathrm{Pb}-206$ to that of $\mathrm{U}-238$ in the sample is found to be 7 .
The value of $\mathbf{P}$ is $\_\_\_\_$ .
[Given: Half-life of U-238 is $4.5 \times 109$ years; $\log _{\mathrm{e}} 2=0.693$ ]