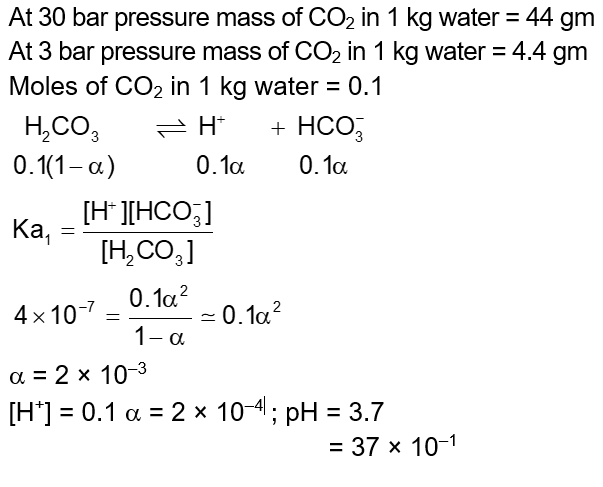A soft drink was bottled with a partial pressure of $\mathrm{CO}_2$ of 3 bar over the liquid at room temperature. The partial pressure of $\mathrm{CO}_2$ over the solution approaches a value of 30 bar when 44 g of $\mathrm{CO}_2$ is dissolved in 1 kg of water at room temperature. The approximate pH of the soft drink is $\_\_\_\_$ $\times 10^{-1}$. (First dissociation constant of $\mathrm{H}_2 \mathrm{CO}_3=4.0 \times 10^{-7}$; $\log 2=0.3 ;$ density of the soft drink $\left.=1 \mathrm{~g} \mathrm{~mL}^{-1}\right)$