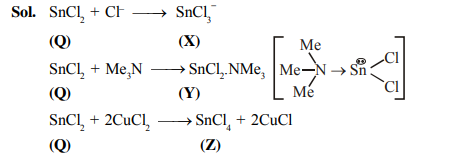A tin chloride $\mathbf{Q}$ undergoes the following reactions (not balanced)
$$
\begin{aligned}
& \mathbf{Q}+\mathrm{Cl}^{-} \rightarrow \mathbf{X} \\
& \mathbf{Q}+\mathrm{Me}_3 \mathrm{~N} \rightarrow \mathbf{Y} \\
& \mathbf{Q}+\mathrm{CuCl}_2 \rightarrow \mathbf{Z}+\mathrm{CuCl}
\end{aligned}
$$
X is a monoanion having pyramidal geometry. Both $\mathbf{Y}$ and $\mathbf{Z}$ are neutral compounds. Choose the correct option(s).