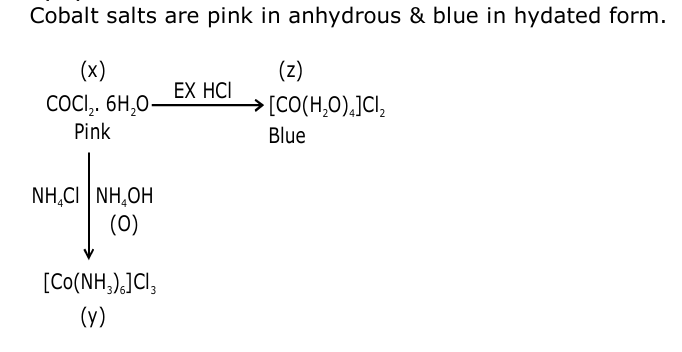
Addition of excess aqueous ammonia to a pink coloured aqueous solution of $\mathrm{MCl}_2 \cdot 6 \mathrm{H}_2 \mathrm{O}(\mathrm{X})$ and $\mathrm{NH}_4 \mathrm{Cl}$ gives an octahedral complex Y in the pressure of air. In aqueous solution, complex Y behaves as 1 : 3 electrolyte. The reaction of $X$ with excess HCl at room temperature results in the formation of a blue coloured compelx Z . The calculated spin only magnetic moment of X and Z is 3.87 B.M., whereas it is zero for complex Y.
Among the following options, which statement(s) is(are) correct ?