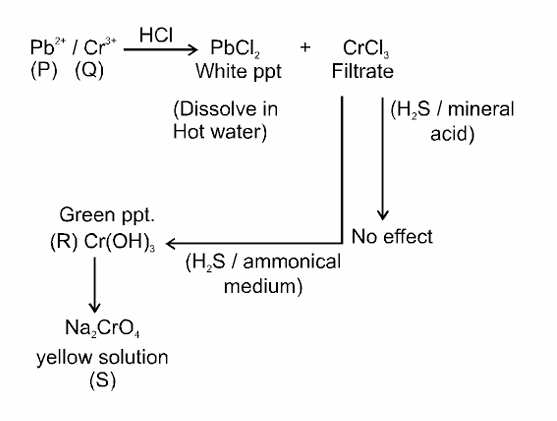
An aqueous solution of a mixture of two inorganic salts, when treated with dilute HCl , gave a precipitate (P) and a filtrate (Q). The precipitate $\mathbf{P}$ was found to dissolve in hot water. The filtrate (Q) remained unchanged, when treated with $\mathrm{H}_2 \mathrm{~S}$ in a dilute mineral acid medium. However, it gave a precipitate $(\mathbf{R})$ with $\mathrm{H}_2 \mathrm{~S}$ in an ammoniacal medium. The precipitate $\mathbf{R}$ gave a coloured solution $(\mathbf{S})$, when treated with $\mathrm{H}_2 \mathrm{O}_2$ in an aqueous NaOH medium.
The coloured solution $\mathbf{S}$ contains