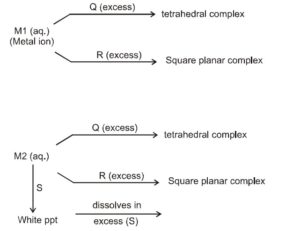
Reaction with ( S ) indicates amphoteric nature of M2 . Amongst the options mentioned for ( S )
[Note : M2 may be $\mathrm{Zn}^{2+}$, which (a) is amphoteric (b) has cordination number 4 and (c) always from tetrahedral complexes. It may be noted that $\mathrm{Be}^{2+}$ will also qualify with these characterstics]
Let us consider the possiblities of M1. M1 should be able to form square planner complex (dsp ${ }^2$ hybridisation) as well as tetrahedral ( $\mathrm{sp}^3$ ). This rules out $\mathrm{Zn}^{2+} \& \mathrm{Cd}^{2+}\left[\because \mathrm{d}^{10}\right.$ configuration will not allow $\mathrm{dsp}^2$ hybridization]
$\mathrm{Ni}^{2+}$ is most suitable
$\mathrm{Ni}^{2+}$ (aq.) $+4 \mathrm{CN}^{-}$(excess) $\rightleftharpoons \mathrm{Ni}(\mathrm{CN})_4^{2-}$ (square planner)
$\mathrm{Ni}^{2+}$ (aq.) $+\mathrm{Cl}^{-}$(excess) $\rightleftharpoons \mathrm{NiCl}_4{ }^{2-}$ (tetrahedral)
[Note : $\mathrm{Co}^{2+}+6 \mathrm{CN}^{-}$(excess) ⟶ $\mathrm{Co}(\mathrm{CN})_6^{4-}$, an octahedral compelex]
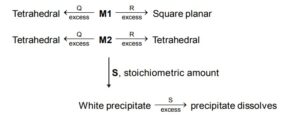 M1, Q and R, respectively are :
M1, Q and R, respectively are : Reaction with ( S ) indicates amphoteric nature of M2 . Amongst the options mentioned for ( S )
Reaction with ( S ) indicates amphoteric nature of M2 . Amongst the options mentioned for ( S )