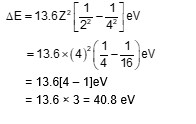An electron of a hydrogen like atom, having $Z=4$, jumps from $4^{\text {th }}$ energy state to $2^{\text {nd }}$ energy state, The energy released in this process, will be: (Given Rch $=13.6 \mathrm{eV}$ ) Where $\mathrm{R}=$ Rydberg constant, $\mathrm{c}=$ Speed of light in vacuum, $\mathrm{h}=$ Planck's constant