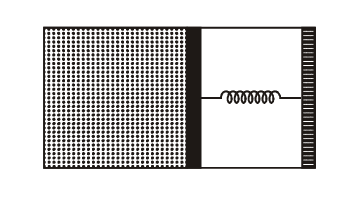
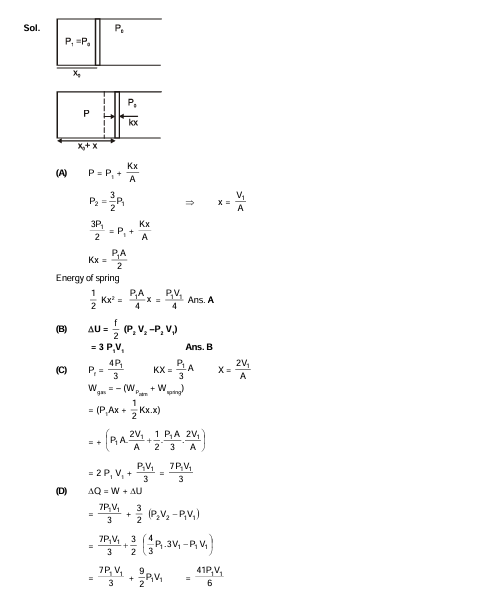
An ideal monoatomic gas is confined in a horizontal cylinder by a spring loaded piston (as shown in the figure). Initially the gas is at temperature $\mathrm{T}_1$, pressure $\mathrm{P}_1$ and volume $\mathrm{V}_1$ and the spring is in its relaxed state. the gas is then heated very slowly to temperature $T_2$, pressure $P_2$ and volume $V_2$. During this process the piston moves out by a distance $x$. Ignoring the friction between the piston and the cylinder, the correct statement(s) is(are)