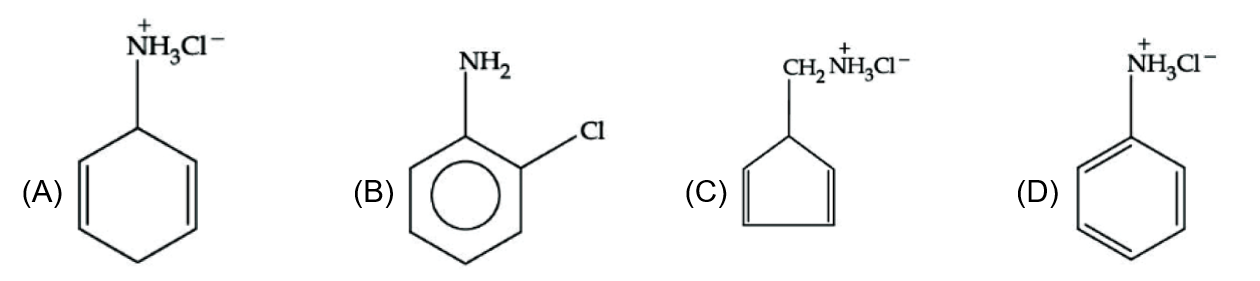
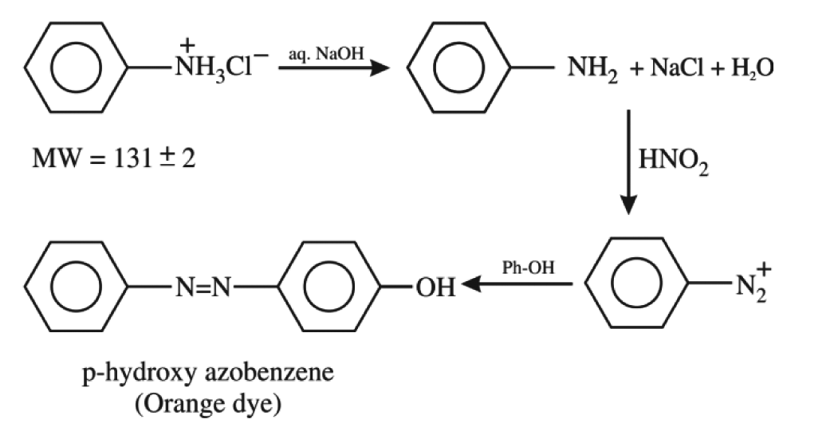
An organic compound 'A' contains nitrogen and chlorine. It dissolves readily in water to give a solution that turns litmus red. Titration of compound 'A' with standard base indicates that the molecular weight of 'A' is 131 ± 2. When a sample of 'A' is treated with aq. NaOH, a liquid separates which contains N but not Cl. Treatment of the obtained liquid with nitrous acid followed by phenol gives orange precipitate. The compound 'A' is :