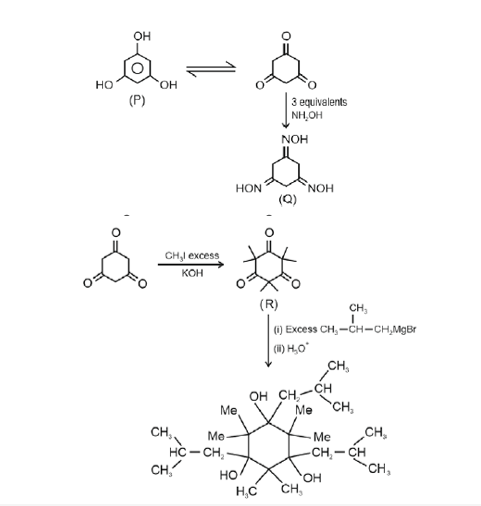
An organic compound $\mathbf{P}$ having molecular formula $\mathrm{C}_6 \mathrm{H}_6 \mathrm{O}_3$ gives ferric chloride test and does not have intramolecular hydrogen bond. The compound $\mathbf{P}$ reacts with 3 equivalents of $\mathrm{NH}_2 \mathrm{OH}$ to produce oxime $\mathbf{Q}$. Treatment of $\mathbf{P}$ with excess methyl iodide in the presence of KOH produces compound $\mathbf{R}$ as the major product.
Reaction of $\mathbf{R}$ with excess iso-butylmagnesium bromide followed by treatment with $\mathrm{H}_3 \mathrm{O}+$ gives compound $\mathbf{S}$ as the major product.
The total number of methyl $\left(-\mathrm{CH}_3\right)$ group(s) in compound $\mathbf{S}$ is $\_\_\_\_$ .