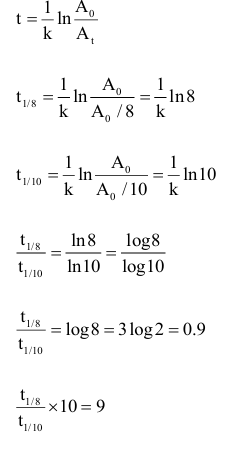An organic compound undergoes first order decomposition. The time taken for decomposition to $\left(\frac{1}{8}\right)^{\text {th }}$ and $\left(\frac{1}{10}\right)^{\text {th }}$ of its initial concentration are $t_{1 / 8}$ and $t_{1 / 10}$ respectively.
What is the value of $\frac{\mathrm{t}_{1 / 8}}{\mathrm{t}_{1 / 10}} \times 10$ ?
$(\log 2=0.3)$