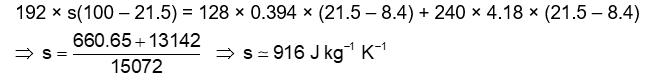An unknown metal of mass 192 g heated to a temperature of 100°C was immersed into a brass calorimeter of mass 128 g containing 240 g of water at a temperature of 8.4°C. Calculate the specific heat of the unknown metal if water temperature stabilizes at 21.5°C. (Specific heat of brass is 394 J $\left.\mathrm{kg}^{-1} \mathrm{~K}^{-1}\right)$