Answer the following by appropriately matching the lists based on the information given in the paragraph.
In a thermodynamics process on an ideal monatomic gas, the infinitesimal heat absorbed by the gas is given by $\mathrm{T} \Delta \mathrm{X}$, where T is temperature of the system and $\Delta \mathrm{X}$ is the infinitesimal change in a thermodynamic quantity $X$ of the system. For a mole of monatomic ideal gas
$\mathrm{X}=\frac{3}{2} \mathrm{R} \ln \left(\frac{\mathrm{T}}{\mathrm{T}_{\mathrm{A}}}\right)+\mathrm{R} \ln \left(\frac{\mathrm{V}}{\mathrm{V}_{\mathrm{A}}}\right)$. Here, R is gas constant, V is volume of gas, $\mathrm{T}_{\mathrm{A}}$ and $\mathrm{V}_{\mathrm{A}}$ are constants.
The List-I below gives some quantities involved in a process and List-II gives some possible values of these quantities.

If the process carried out on one mole of monatomic ideal gas is as shown in figure in the PV-diagram with $P_0 V_0=\frac{1}{3} R T_0$, the correct match is,
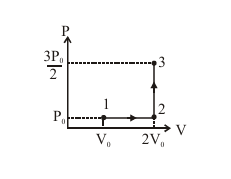
 If the process carried out on one mole of monatomic ideal gas is as shown in figure in the PV-diagram with $P_0 V_0=\frac{1}{3} R T_0$, the correct match is,
If the process carried out on one mole of monatomic ideal gas is as shown in figure in the PV-diagram with $P_0 V_0=\frac{1}{3} R T_0$, the correct match is,

