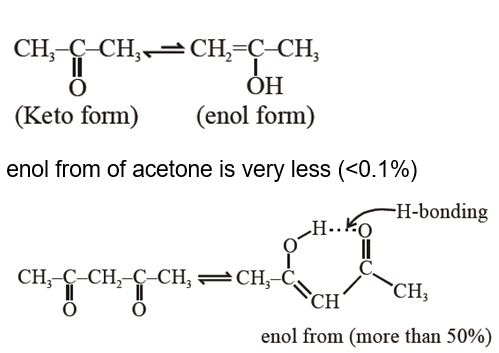
Assertion A : Enol form of acetone $\left[\mathrm{CH}_3 \mathrm{COCH}_3\right]$ exists in $<0.1 \%$ quantity. However, the enol form of acetyl acetone $\left[\mathrm{CH}_3 \mathrm{COCH}_2 \mathrm{OCCH}_3\right]$ exists in approximately $15 \%$ quantity.
Reason R : enol form of acetyl acetone is stabilized by intramolecular hydrogen bonding, which is not possible in enol form of acetone. Choose the correct statement :