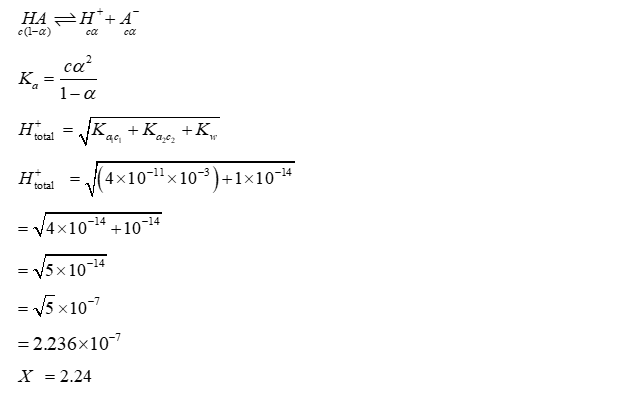At $25^{\circ} \mathrm{C}$, the concentration of $\mathrm{H}^{+}$ions in $1.00 \times 10^{-3} \mathrm{M}$ aqueous solution of a weak monobasic acid having acid dissociation constant $\left(K_a\right)$ of $4.00 \times 10^{-11}$ is $X \times 10^{-7} M$.
The value of $X$ is $\_\_\_\_$
Use: Ionic product of water $\left(K_w\right)=1.00 \times 10^{-14}$ at $25^{\circ} \mathrm{C}$