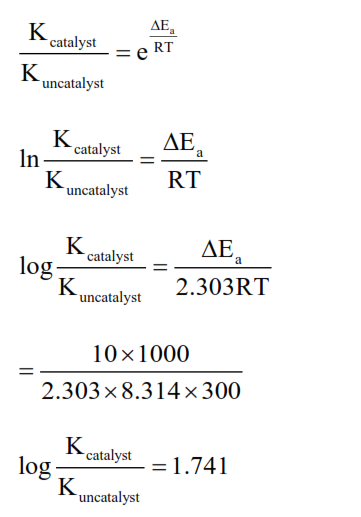
At $27^{\circ} \mathrm{C}$ in presence of a catalyst, activation energy of a reaction is lowered by $10 \mathrm{~kJ} \mathrm{~mol}^{-1}$. The logarithm of ratio of $\frac{\mathrm{k} \text { (catalysed) }}{\mathrm{k} \text { (uncatalysed) }}$ is....
(Consider that the frequency factor for both the reactions is same)