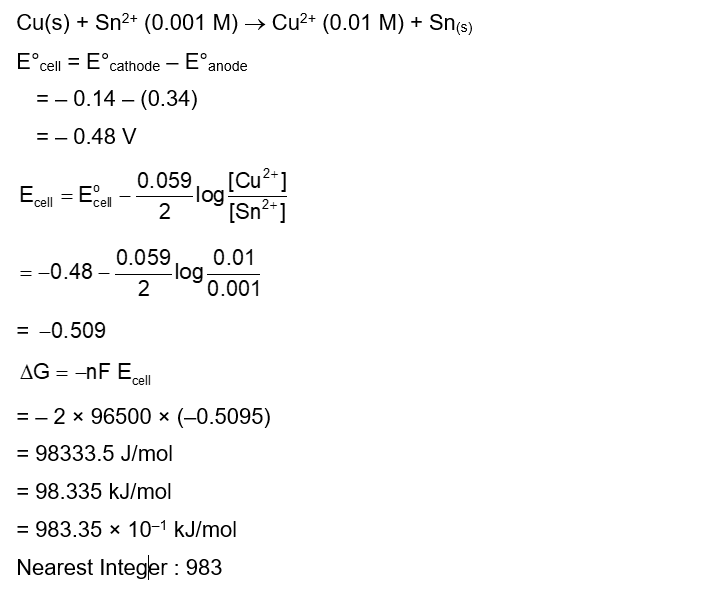
$$
\mathrm{Cu}(\mathrm{~s})+\mathrm{Sn}^{2+}(0.001 \mathrm{M}) \rightarrow \mathrm{Cu}^{2+}(0.01 \mathrm{M})+\mathrm{Sn}(\mathrm{~s})
$$
The Gibbs free energy change for the above reaction at 298 K is $\mathrm{x} \times 10^{-1} \mathrm{~kJ} \mathrm{~mol}^{-1}$;
The value of $x$ is $\_\_\_\_$ . [nearest integer]
$\left[\right.$ Given : $\left.\mathrm{E}_{\mathrm{Cu}^{2+} / \mathrm{Cu}}^{\ominus}=0.34 \mathrm{~V} ; \mathrm{E}_{\mathrm{Sn}^{2+} / \mathrm{Sn}}^{\ominus}=-0.14 \mathrm{~V} ; \mathrm{F}=96500 \mathrm{Cmol}^{-1}\right]$