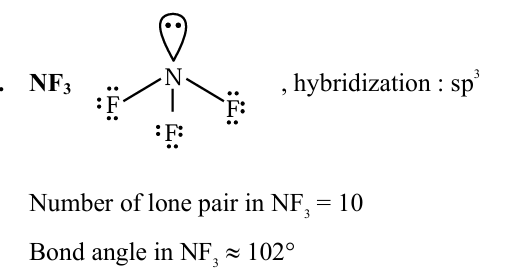
Identify the molecule $(\mathrm{X})$ with maximum number of lone pairs of electrons (obtained using Lewis dot structure) among $\mathrm{HNO}_3, \mathrm{H}_2 \mathrm{SO}_4, \mathrm{NF}_3$ and $\mathrm{O}_3$. Choose the correct bond angle made by the central atom of the molecule $(\mathrm{X})$.