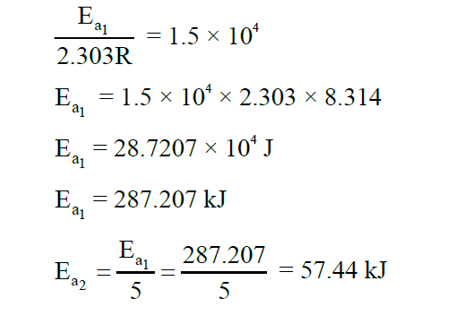Consider $A \xrightarrow{k_1} B$ and $C \xrightarrow{k_2} D$ are two reactions. If the rate constant $\left(k_1\right)$ of the $A \longrightarrow B$ reaction can be expressed by the following equation $\log _{10} \mathrm{k}=14.34-\frac{1.5 \times 10^4}{\mathrm{~T} / \mathrm{K}}$ and activation energy of $C \longrightarrow D$ reaction $\left(E a_2\right)$ is $\frac{1}{5}$ th of the $A \longrightarrow B$ reaction $\left(E a_1\right)$, then the value of $\left(E a_2\right)$ is __ $\mathrm{kJmol}^{-1}$. (Nearest Integer)