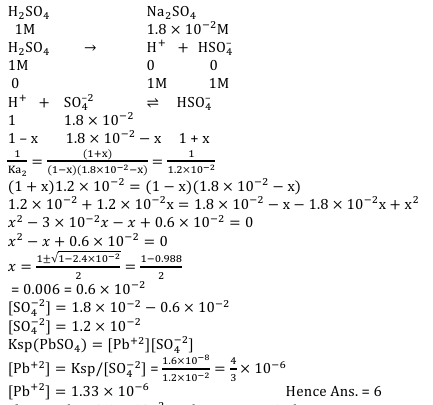Concentration of $\mathrm{H}_2 \mathrm{SO}_4$ and $\mathrm{Na}_2 \mathrm{SO}_4$ in a solution is 1 M and $1.8 \times 10^{-2} \mathrm{M}$, respectively. Molar solubility of $\mathrm{PbSO}_4$ in the same solution is $\mathrm{X} \times 10^{-\mathrm{Y}} \mathrm{M}$ (expressed in scientific notation). The value of Y is $\_\_\_\_$ .
[Given: Solubility product of $\mathrm{PbSO}_4\left(\mathrm{~K}_{\mathrm{sp}}\right)=1.6 \times 10^{-8}$. For $\mathrm{H}_2 \mathrm{SO}_4, \mathrm{~K}_{\mathrm{a} 1}$ is very large and $\left.\mathrm{K}_{\mathrm{a} 2}=1.2 \times 10^{-2}\right]$