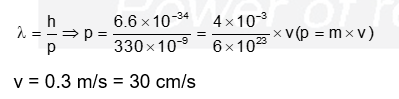Consider a helium (He) atom that absorbs a photon of wavelength 330 nm . The change in the velocity (in $\mathrm{cm} \mathrm{s}^{-1}$ ) of He atom after the photon absorption is $\_\_\_\_$ .
(Assume: Momentum is conserved when photon is absorbed.
Use: Planck constant $=6.6 \times 10^{-34} \mathrm{~J} \mathrm{~s}$, Avogadro number $=6 \times 10^{23} \mathrm{~mol}^{-1}$, Molar mass of $\left.\mathrm{He}=4 \mathrm{~g} \mathrm{~mol}^{-1}\right)$