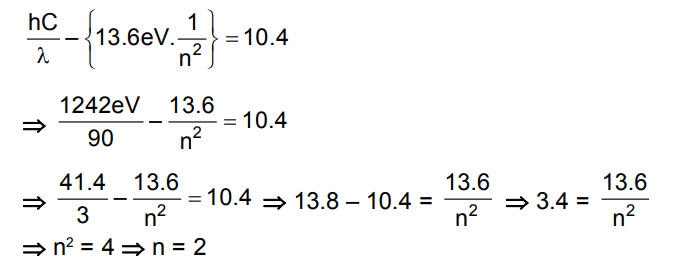Consider a hydrogen atom with its electron in the $\mathrm{n}^{\text {th }}$ orbital. An electromagnetic radiation of wavelength 90 nm is used to ionize the atom. If the kinetic energy of the ejected electron is 10.4 eV , then the value of $n$ is $(\mathrm{hc}=1242 \mathrm{eV} \mathrm{nm})$