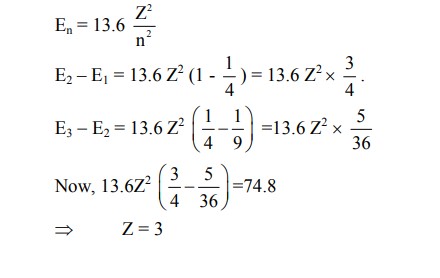Consider a hydrogen-like ionized atom with atomic number Z with a single electron. In the emission spectrum of this atom, the photon emitted in the $n=2$ to $n=1$ transition has energy 74.8 eV higher than the photon emitted in the $\mathrm{n}=3$ to $\mathrm{n}=2$ transition. The ionization energy of the hydrogen atom is 13.6 eV . The value of Z is $\_\_\_\_$ .