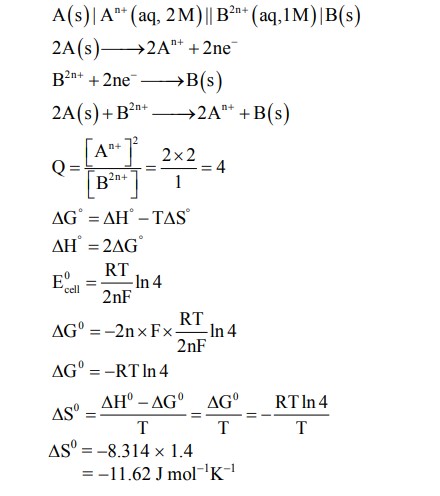Consider an electrochemical cell: $\mathrm{A}(\mathrm{s})\left|\mathrm{A}^{\mathrm{n}+}(\mathrm{aq}, 2 \mathrm{M}) \| \mathrm{B}^{2 \mathrm{n}^{+}}(\mathrm{aq}, 1 \mathrm{M})\right| \mathrm{B}(\mathrm{s})$. The value of $\Delta \mathrm{H}^{\ominus}$ for the cell reaction is twice that of $\Delta \mathrm{G}^{\ominus}$ at 300 K . If the emf of the cell is zero, the $\Delta \mathrm{S}^{\ominus}$ (in $\mathrm{J} \mathrm{K}^{-1} \mathrm{~mol}^{-1}$ ) of the cell reaction per mole of B formed at 300 K is $\_\_\_\_$ .
(Given: $\ln (2)=0.7, \mathrm{R}$ (universal gas constant) $=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1} . H, S$ and $G$ are enthalpy, entropy and Gibbs energy, respectively.)