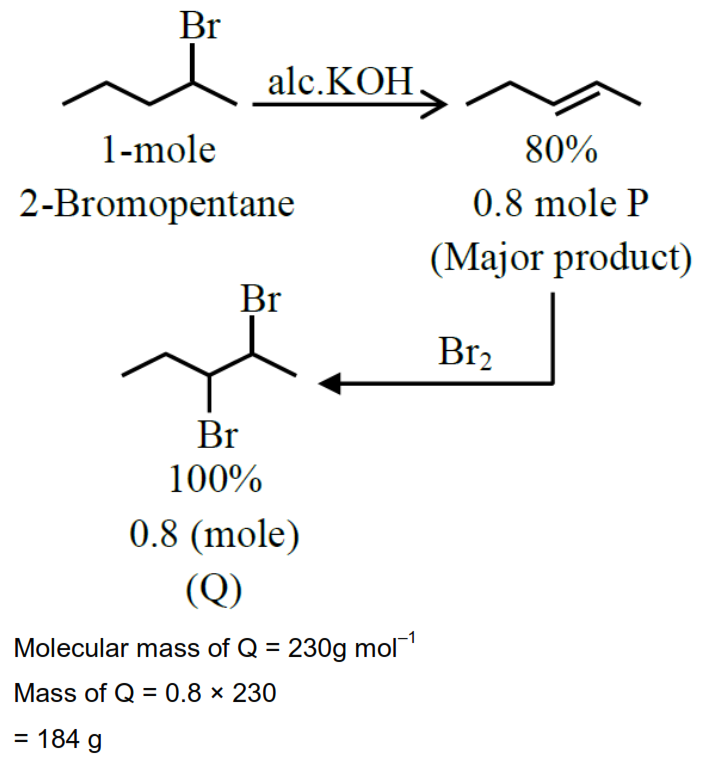
Consider the above sequence of reactions. 151 g of 2-bromopentane is made to react. Yield of major product P is $80 \%$ whereas Q is $100 \%$.
Mass of product Q obtained is $\_\_\_\_$ g.
(Given molar mass in $\mathrm{gmol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16, \mathrm{Br}: 80$ )