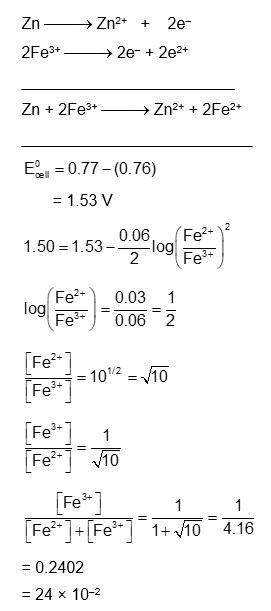Consider the cell at $25^{\circ} \mathrm{C} \mathrm{Zn}\left|\mathrm{Zn}^{2+}(\mathrm{aq}),(1 \mathrm{M}) \| \mathrm{Fe}^{3+}(\mathrm{aq}), \mathrm{Fe}^{2+}(\mathrm{aq})\right| \mathrm{Pt}(\mathrm{s})$
The fraction of total iron present as $\mathrm{Fe}^{3+}$ ion at the cell potential of 1.500 V is $\mathrm{x} \times 10^{-2}$. The value of x is
$\_\_\_\_$ (Nearest integer)
$\left(\right.$ Given : $\left.\mathrm{E}_{\mathrm{Fe}^{3+} / \mathrm{Fe}^{2+}}^0=0.77 \mathrm{~V}, \mathrm{E}_{\mathrm{Zn}^{2+} / \mathrm{Zn}}^0=-0.76 \mathrm{~V}\right)$