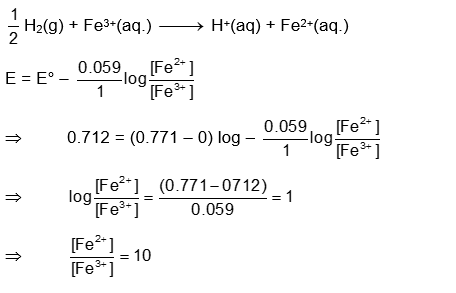Consider the cell
$
\operatorname{Pt}(\mathrm{s})|\mathrm{H}(\mathrm{~s})(1 \mathrm{~atm})| \mathrm{H}^{+}\left(\mathrm{aq},\left[\mathrm{H}^{+}\right]=1\right) \| \mathrm{Fe}^{3+}(\mathrm{aq}), \mathrm{Fe}^{2+}(\mathrm{aq}) \mid \operatorname{Pt}(\mathrm{s})
$
Given: $\mathrm{E}_{\mathrm{Fe}^{3+} / \mathrm{Fe}^{2+}}^{\infty}=0.771 \mathrm{~V}$ and $\mathrm{E}_{\mathrm{H}^{+} / \frac{1}{2} \mathrm{H}_2}^{\infty}=0 \mathrm{~V}, \mathrm{~T}=298 \mathrm{~K}$
If the potential of the cell is 0.712 V the ratio of concentration of $\mathrm{Fe}^{2+}$ to $\mathrm{Fe}^{2+}$ is $\_\_\_\_$ (Nearest integer)