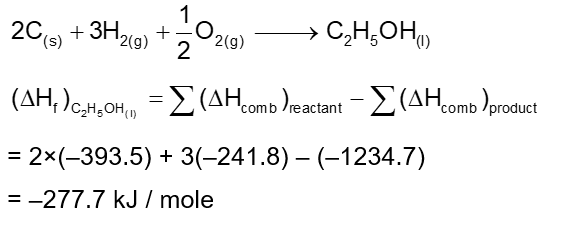Consider the following data
Heat of combustion of $\mathrm{C}(\mathrm{s})=-393.5 \mathrm{~kJ} \mathrm{~mol}^{-1}$
Heat of combustion of $\mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}(\mathrm{I})=-1234.7 \mathrm{~kJ} \mathrm{~mol}^{-1}$
The heat of formation of $\mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}(\mathrm{I})$ is (-) $\_\_\_\_$ $\mathrm{kJ} \mathrm{mol}^{-1}$ (Nearest Integer)