Consider the following reaction.
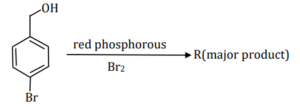
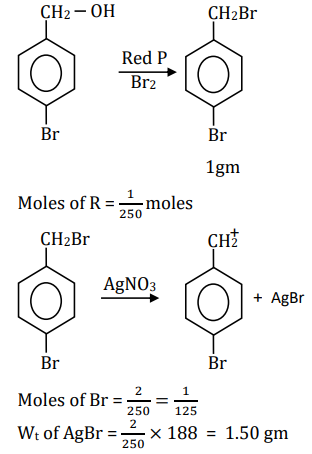
On estimation of bromine in 1.00 g of $\mathbf{R}$ using Carius method, the amount of AgBr formed (in g ) is $\_\_\_\_$ [Given: Atomic mass of $\mathrm{H}=1, \mathrm{C}=12, \mathrm{O}=16, \mathrm{P}=31, \mathrm{Br}=80, \mathrm{Ag}=108$ ]