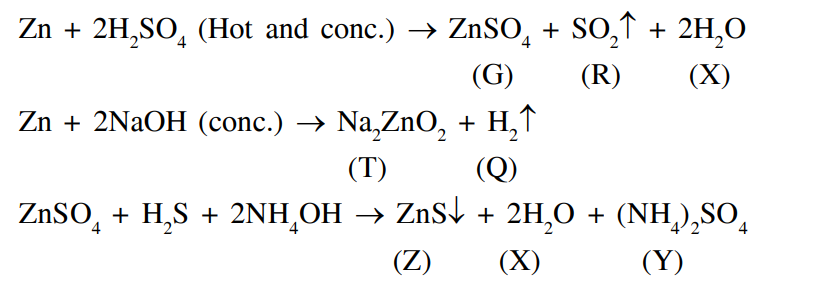Consider the following reactions (unbalanced)
$$
\begin{aligned}
& \mathrm{Zn}+\text { hot conc. } \mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{G}+\mathrm{R}+\mathrm{X} \\
& \mathrm{Zn}+\text { conc. } \mathrm{NaOH} \rightarrow \mathrm{~T}+\mathrm{Q} \\
& \mathrm{G}+\mathrm{H}_2 \mathrm{~S}+\mathrm{NH}_4 \mathrm{OH} \rightarrow \mathrm{Z} \text { ( a precipitate) }+\mathrm{X}+\mathrm{Y}
\end{aligned}
$$
Choose the correct option(s).