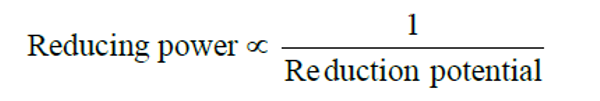Consider the following reduction processes :
$$
\begin{aligned}
& \mathrm{Al}^{3+}+3 \mathrm{e}^{-} \longrightarrow \mathrm{Al}(\mathrm{~s}), \mathrm{E}^0=-1.66 \mathrm{~V} \\
& \mathrm{Fe}^{3+}+\mathrm{e}^{-} \longrightarrow \mathrm{Fe}^{2+}, \mathrm{E}^0=+0.77 \mathrm{~V} \\
& \mathrm{Co}^{3+}+\mathrm{e}^{-} \longrightarrow \mathrm{Co}^{2+}, \mathrm{E}^0=+1.81 \mathrm{~V} \\
& \mathrm{Cr}^{3+}+3 \mathrm{e}^{-} \longrightarrow \mathrm{Cr}(\mathrm{~s}), \mathrm{E}^0=-0.74 \mathrm{~V}
\end{aligned}
$$
The tendency to act as reducing agent decreases in the order :