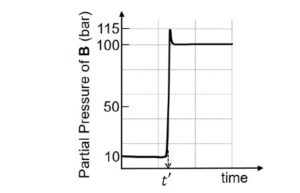
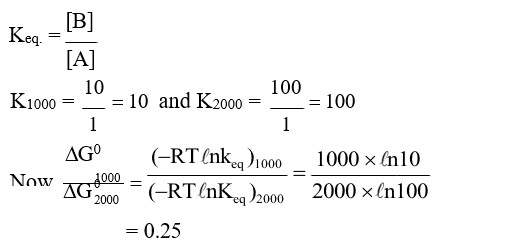Consider the reaction $\mathbf{A} \rightleftharpoons \mathbf{B}$ at 1000 K . At time $\mathrm{t}^{\prime}$, the temperature of the system was increased to 2000 K and the system was allowed to reach equilibrium. Throughout this experiment the partial pressure of $\mathbf{A}$ was maintained at 1 bar. Given below is the plot of the partial pressure of $\mathbf{B}$ with time. What is the ratio of the standard Gibbs energy of the reaction at 1000 K to that at $2000 \mathrm{~K} ?$