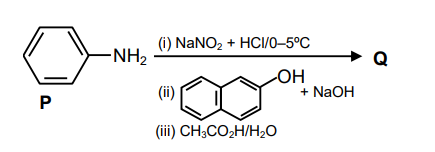
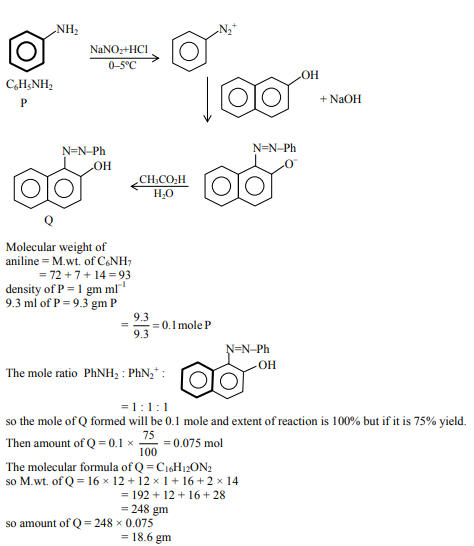
Consider the reaction sequence from $\mathbf{P}$ to $\mathbf{Q}$ shown below. The overall yield of the major product $\mathbf{Q}$ from $\mathbf{P}$ is $75 \%$. What is the amount in grams of $\mathbf{Q}$ obtained from 9.3 mL of $\mathbf{P}$ ? (Use density of $\mathbf{P}=1.00 \mathrm{~g} \mathrm{~mL}^{-1}$, Molar mass of $\mathrm{C}=12.0, \mathrm{H}=1.0, \mathrm{O}=16.0$ and $\left.\mathrm{N}=14.0 \mathrm{~g} \mathrm{~mol}^{-1}\right)$