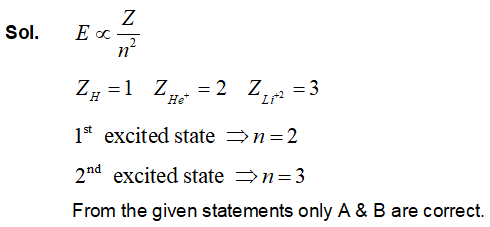Considering Bohr's atomic model for hydrogen atom:
(A) the energy of H atom in ground state is same as energy of $H{e^ + }$ ion in its first excited state.
(B) the energy of H atom in ground state is same as that for $L{i^{ + + }}$ ion in its second excited state.
(C) the energy of H atom in its ground state is same as that of $H{e^ + }$ ion for its ground state.
(D) the energy of $H{e^ + }$ ion in its first excited state is same as that for $L{i^{ + + }}$ ion in its ground state.
Choose the correct answer from the options given below :