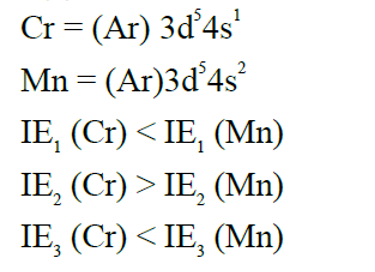Given below are two statements :
Statement I: The first ionization enthalpy of Cr is lower than that of Mn .
Statement II: The second and third ionization enthalpies of Cr are higher than those of Mn . In the light of the above statements, choose the correct answer from the options given below :