Dilution processes of different aqueous solutions, with water, are given in LIST-I. The effects of dilution of the solutions on [ $\mathrm{H}^{+}$] are given in LIST-II.
(Note: Degree of dissociation $(\alpha)$ of weak acid and weak base is $\ll 1$; degree of hydrolysis of salt $\ll 1$; $\left[\mathrm{H}^{+}\right]$represents the concentration of $\mathrm{H}^{+}$ions)
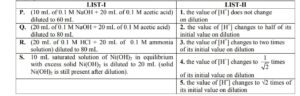
Match each process given in LIST-I with one or more effect(s) in LIST-II. The correct option is
 Match each process given in LIST-I with one or more effect(s) in LIST-II. The correct option is
Match each process given in LIST-I with one or more effect(s) in LIST-II. The correct option is