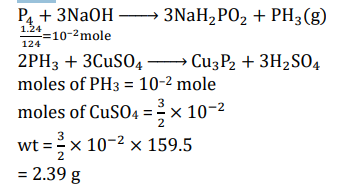Dissolving 1.24 g of white phosphorous in boiling NaOH solution in an inert atmosphere gives a gas Q . The amount of $\mathrm{CuSO}_4$ (in g ) required to completely consume the gas Q is $\_\_\_\_$ .
[Given: Atomic mass of $\mathrm{H}=1, \mathrm{O}=16, \mathrm{Na}=23, \mathrm{P}=31, \mathrm{~S}=32, \mathrm{Cu}=63$ ]